Your cosmetics, our project
Are you looking for a partner to support you at every stage of developing your cosmetic products?
Our lab can assist you

Formulation

Manufacturing

Filling

Packaging

Logistics
Much more than a cosmetics lab: a global cosmetics hub
We are ISO 22716 (Good Manufacturing Practices) and ISO 13485 (Medical Device) certified, which guarantees that your cosmetic products will be developed in accordance with European standards and Good Manufacturing Practices.
You benefit from our extensive expertise and more than 15 years of experience to meet any needs you may have:
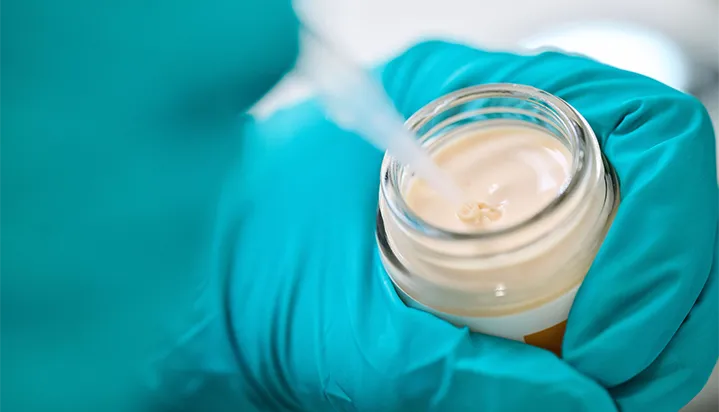
Comprehensive expertise
in cosmetic products: face and body products, hygiene, care products for men, baby care and natural and organic care products

Large production capacity
from 1,000 units to millions
15,000 m2 production area

Bespoke cosmetics packaging
all kinds of containers, packaging and formats, including tubes, bottles, jars and sachets

Point-of-sale advertising
Creation of bespoke displays and advertising media for cosmetic products

Full service
Expertise across the value chain, from cosmetic formulation to cosmetic filling and packaging

Benefits
Speed, flexibility and responsiveness
Regardless of the form your cosmetic products take, or the kind of packaging you choose, our company is flexible and boasts the equipment be able to adapt to what you’re looking for.
Our cosmetics lab in figures
international clients (both new and established brands)
million units produced per year
filling and packaging lines
employees
years of experience
manufacturing tanks, ranging in size from 5 kg to 3 T
m2 of storage
What customers have to say about our cosmetics lab


Our CONPALUX support guide
This document is completely FREE
"*" indicates required fields